The company
Manufacturers since 1973
Our Mission
To look after people’s health by developing reliable medical technology.
Established in 1973 by one of the pioneers of the sector in Spain, Benjamín Herranz, HERSILL has extensive experience as a manufacturer of medical equipment.
Our eagerness for technological innovation, continuous improvement, customer service, and the tireless work of many years, has allowed us to position ourselves solidly in the competitive and demanding world market of medical devices.
We design and manufacture devices of the highest quality and with the most modern technology, making them available to medical and health professionals, helping them to treat ailments and take care of people’s health.
We help to maintain in the long term the prestige and invaluable trust that health professionals place in our company.
We provide maximum benefit to the health of patients treated with our products.
We promote the satisfaction and prosperity, both economically and profesionally and personally, of our customers and suppliers, as well as the staff working at HERSILL and its partners.
To face this challenge, HERSILL counts on an excellent team of professionals and collaborators, depositaries of an extensive know-how and competence in the performance of its mission, working together with the same mission and vision, under a single premise, OUR VALUES.
Responsibility. We meet commitments and develop the skills to do so.
Eagerness. We feel involved in a common project: achieving our VISION.
Excellence. We work with a criterion of continuous improvement.
HERSILL FULL RANGE OF PRODUCTS – CATALOGUE
50 years of innovation
PRESS AREA

Our R+D
Innovation is the pillar of a knowledge-based society, and is also fundamental to the dynamism of a company.
HERSILL’s R+D Department enjoys recognized prestige among companies in the sector and surrounding universities and institutions. Our work focuses on positioning and maintaining HERSILL at the forefront of health technologies, and our activity culminates in the development of new products, which allow HERSILL to remain among the elite of the sector at an international level.
The R+D Department is located in modern facilities composed of offices, development laboratory and testing and metrology laboratory, annexed to the HERSILL factory in Móstoles. It integrates a R+D exclusively dedicated, multidiscipinary team, made up of engineers with expertise in healthcare technology and doctors in medicine, and has collaboration agreements with hospitals and universities.
We are highly aware of the importance of the exchange of knowledge between the entire Organization, and therefore the R+D Department is not an isolated entity, but, in addition to dealing with the transfer of technology to the production plant, collaborates directly with the commercial sales and marketing teams, both our own and those of our clients, in the prospection of trends and the delivery of solutions that allow us to outperform the market.

Our Quality
HERSILL applies to all its areas of work, a high level of quality and exigency, guaranteed by the main international quality certifications, keeping them always in force.
From product conception to after-sales service and customer service, including technological development, manufacturing and marketing.
It is all part of a set of processes designed to achieve our main objective: the full satisfaction of our customers and users of our medical products.

Our Corporate Social Responsibility
At HERSILL we are strongly committed to the 2030 Sustainable Development Goals.
- Environmental policy, certified according to ISO 14 001:2015 standard.
- Our photovoltaic system produces more than half of the energy we consume.
- We collaborate with the Food Bank of Madrid and with the Red Cross of Mostoles, Madrid (Spain). We also work closely with Special Employment Centres, integrating people with disabilities in key tasks of our activity.
- We help projects of international NGOs such as UNICEF.
- We collaborate with various foundations, such as the Recover Foundation.

OUR Export
HERSILL distributes its medical equipment through its network of expert representatives in the sale of medical equipment with presence in more than 100 countries.
Earning the trust of the market during these years and keeping it day by day, is the result of our strong customer orientation and the willingness to cooperate flexibly and constructively with them.

Our History

1973
HERSILL, S.L. was founded in Madrid, the 22th of February 1973 by Benjamín Herranz Escamilla, pioneer in manufacturing and development of medical equipments in Spain.

1976
First facilities in Villaviciosa de Odón (Madrid), 67 m2 surface area (office, workshop and warehouse). Launch of first HERSILL in-house manufactured range of products: pressure regulators, flowmeters, humidifiers and, Venturi-type suction device onto a portable cart.




1980
HERSILL moves to new bigger Villaviciosa de Odón facilities, 250 m2 surface area. New Venturi-type suction system. Launch of first rail-based systems.




1984
HERSILL moves to the first Móstoles industrial unit (Madrid), 1,000 m2 surface area. New flow meter design.

1985
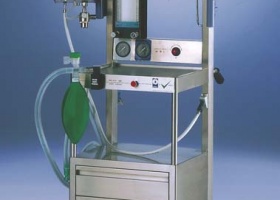
ALERTA 2000 Oxygen and protoxide control supply anaesthetic system. First portable anaesthesia machine.



1991
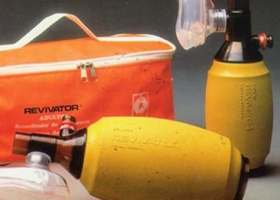
Producing means update, implementing CNC machining (computer numerical control). First REVIVATOR® manual resuscitator. OXIVAC® suitcase line extension.





1996
First ball flow meters with selector system. Launch of rotameter line. New pressure gauge flowmeter design.

1997
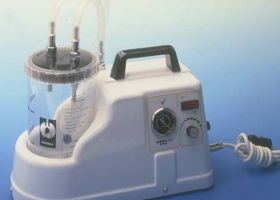
New design of VACUSILL® suction regulators and VACUJET® Venturi-type vacuum systems. First EUROVAC® electrical suction device line for operating rooms.

1998
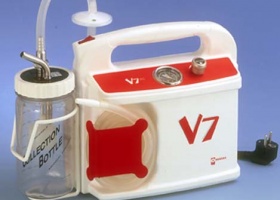
ISO 9002 and CE Marking are granted (quality certificates for medical devices). First VITAE® 2000 anaesthesia pneumatic ventilator. Water column suction device debut. V5 electrical suction pump design upgrade.

1999
HERSILL moves to new built industrial unit and office building in Móstoles (Madrid), surface area 3,500 m2. ORBI anaesthesia portable unit design upgrade and first KAV anaesthesia circular circuit launch.



2003
EN ISO 13488 quality certification for medical products granted. New electronic ventilator for anaesthesia VITAE® PRO.



2006
First PERFLOW® click-style oxygen flowmeter line. First P-300 pressure regulators. New VACUSILL® suction regulator family.

2007
ISO 13485 quality certification for medical products granted. VACUP® suction foot pedal (patent granted). New EUROVAC® H-90 electric surgical suction machine and CAVS emergency cart.

2008
HERSILL is registered as manufacturer with the FDA. PERFLOW® click-style air flowmeters. New OXYLL® ball flowmeters including flow selector piece. New OXIVAC® suitcase models.

2009
Launch of first INGAS oxygen cylinder electronic changeover system for ambulances. New VACUSILL 3 suction regulators for hospital pipelines.



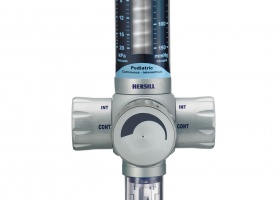
2013
VACUSILL® 3 continuous-intermittent vacuum regulator suitable for subglottic suction and fully autoclavable.




2019
On April 19th Mr. Herranz, founder of HERSILL, passes away. Obtaining the CE mark for the VITAE® 40 ventilator.


2021
The Royal Academy of Engineering awards the Academiae Dilecta Award 2021 to HERSILL for “its vital role in developing and launching products to meet the most critical moments during the coronavirus pandemic.”
Contact HERSILL